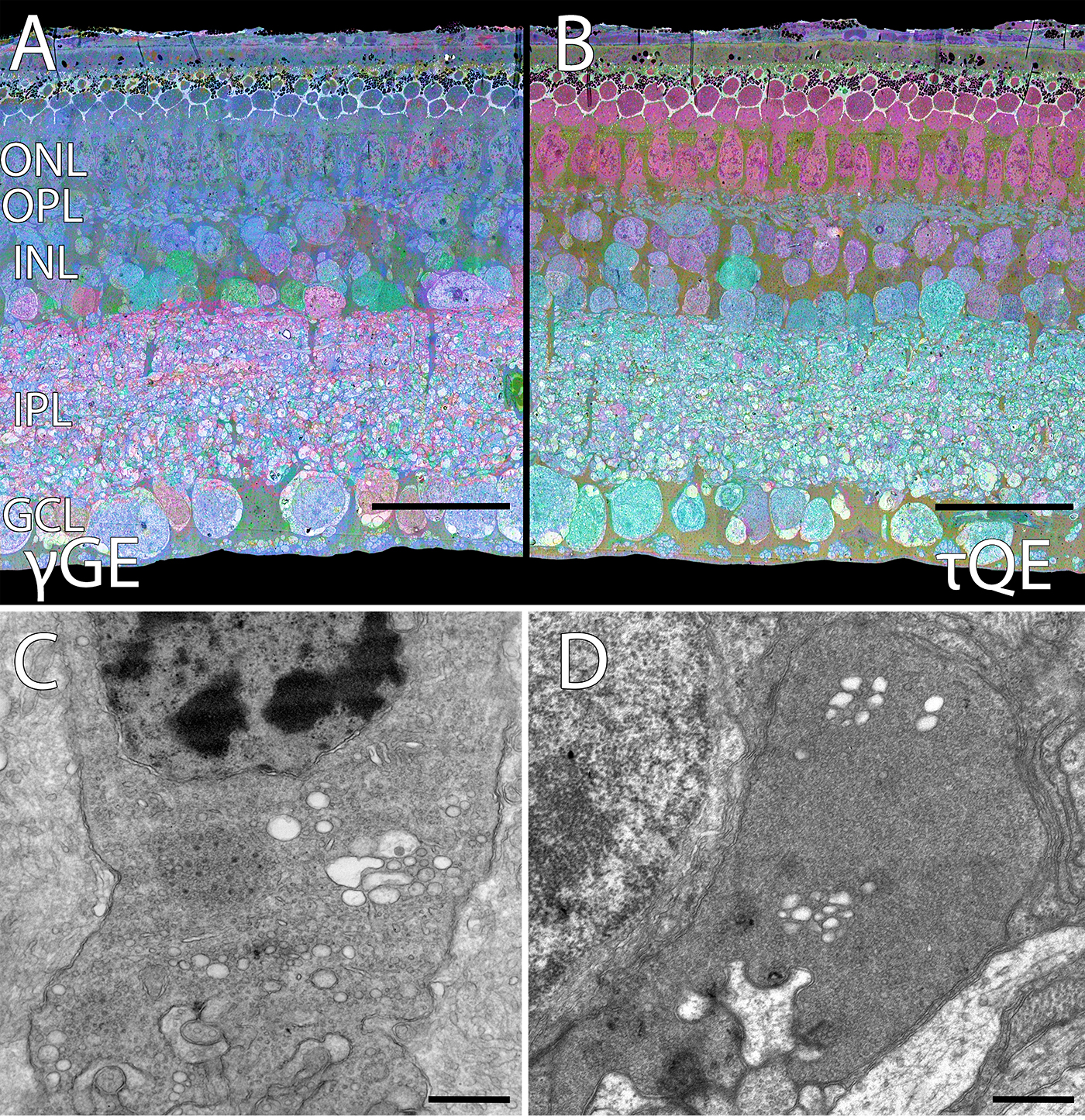
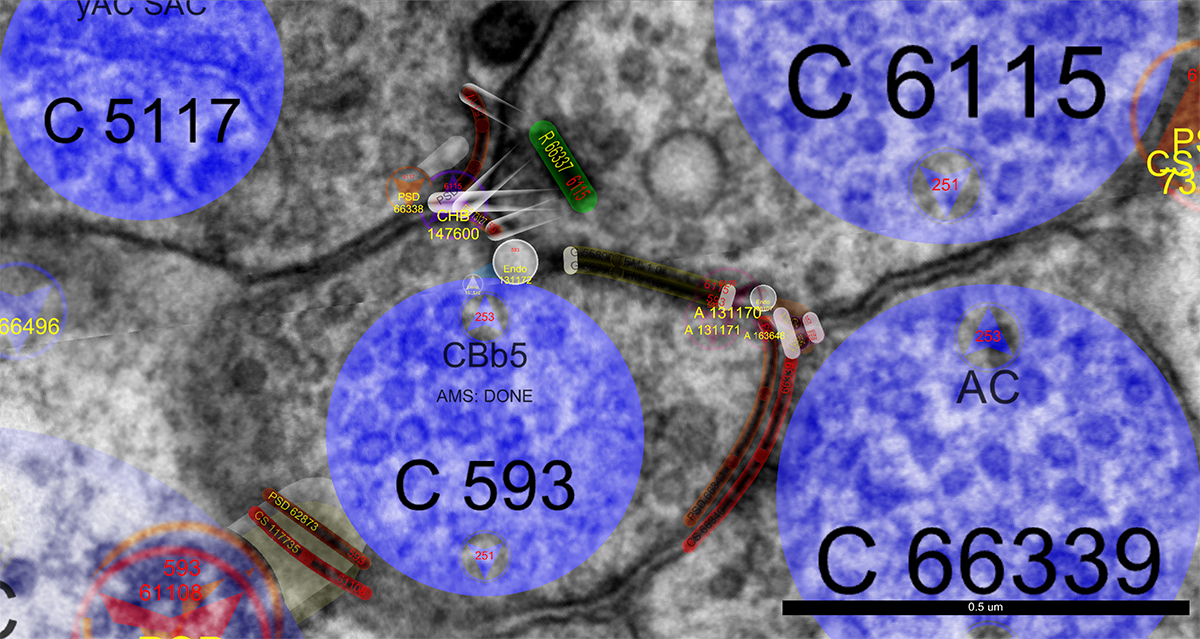
We are pleased to reveal that Dr. Bryan William Jones has been selected for an RPB Stein Innovation Award from Research to Prevent Blindness. This particular project is something that we’ve been scheming for a while and leverages an approach to comparative anatomy to study the ground squirrel retina. The unique thing about the 12-lined ground squirrel retina is that the photoreceptors of this organism degenerate when it hibernates. The outer segments of the photoreceptors degenerate and the synapses that connect them to the first synapse of the visual system dissolves in much the same way as when the retina degenerates in human diseases like retinitis pigmentosa, and age-related macular degeneration. The trick is: When the 13-lined ground squirrel comes out of hibernation, their retinas regenerate and their synapses reconnect giving us an incredible opportunity to explore plasticity in their nervous systems.
Category Archives: Daily
Happy New Year, 2022
“Those of us who survived, are now through the 2nd year of this pandemic.”
That sentence sounds like the introduction to a post apocalyptic story, and something that while not completely unexpected in my lifetime, still has a rawness to it, and an anger to it because of how the world has handled things. Our distraction with political authoritarianism across the world, and an obsession with individual rights over that of society has made this far worse than it otherwise would in a healthy global society. We are now looking at over 800,000 Americans are dead from COVID, with over 5.4 million dead worldwide, and we are not yet done. If you had told me that these would be the numbers back in the first week of March 2020 when I shut the lab down to protect everyone in it, I might not have believed you.
We started the year 2021 in the lab with a chiller for one of our electron microscopes that failed. That took out half our electron microscopy infrastructure for a period of time, until we could get it back up and running, which of course was complicated by the pandemic. I’m grateful for Mark Kirkpatrick, and Kevin Mcilwrath from JEOL @JEOLUSA who helped us maintain the microscopes and keep them up and running. 2021 continued the hits with lots of new bureaucracy initiatives at the University of Utah with IT, and HR, and purchasing, and travel, that are an incredible tax on time and resources. If I had enough resources to help balance out those demands, it would be acceptable, but as it is, I’m getting further and further away from the science, which is seriously bumming me out. I got into this game to dig deep into the science. This skyrocketing administrative burden on top of all the other things that I’ve had to deal with as a result of the pandemic has really stressed things out. Yet we’ve still been incredibly productive through the pandemic, mostly because of the work of personnel in the lab, Jia-Hui, Jamie, Nat @NatQuayleNelson, Crystal @CSigulinsky, Becca @BeccaPfeiffer19, Jeebika, Selena, Olivia, Isabel and Gabe, all of whom I am immensely grateful for.
This year, despite the 2nd year of a global pandemic, we’ve still managed to publish a book chapter, 6 manuscripts, 2 abstracts, participated in teaching new faculty at the University of Utah School of Medicine, co-organized the Moran Eye Center Translational Research Day with Leah Owen, started faculty hiring initiatives, reviewed grants, started a new retinal connectomics initiative that will meet for the first time in January, submitted a grant renewal (triaged), submitted three other grants (not funded), taught classes for grad students, started serving on 3 PhD student committees, brought a couple of new undergraduates into the lab, had one undergraduate win Outstanding Undergraduate Researcher of The Year Award, had postdocs present at a couple of meetings, had a couple of PhD students rotate through the lab, gave 5 invited talks, initiated two more collaborations and more, all while struggling through unprecedented difficulties related to the pandemic.
The New Year 2022 looks promising, and we hope for a slow normalization of things. January will bring a new technician to the lab, and the first meeting of a select group of people for a new retinal connectomics initiative that we’ll be rolling out to a wider audience ASAP. We’ll also have a new collaborative manuscript that has just been accepted in Nature. Later in the first part of the year, we’ll have another three manuscripts submitted, a grant renewal that will go in, and we might have a new PhD student joining the lab as well as a new undergraduate. Hopefully later in the year, some of the irons that I have in the fire will return on those investments, and we’ll start to normalize some other things like meetings. ARVO is planning on being a hybrid meeting, and I’ve agreed to a special interest group presentation there with Michael Redmond. We’ll see what other meetings happen next year, pending outcomes from the pandemic. I’d love to be able to send the postdocs to meetings this year. ISER was pushed out to February of 2023 because of the pandemic, so that won’t be happening this year as was previously planned. Further out in the year is harder to predict, but we should be finished with our third pathoconnectome, as well as our primate retinal connectome, paving the way for us to begin building our two human pathoconnectomes.
However this year ends up, we here at the Marclab for Connectomics wish you a Happy New Year for 2022, and much success and happiness.
Jessica Garcia White Coat Ceremony
Seminar: Retinas, How Are They Wired? How Does Wiring Change In Disease?
PI, Bryan William Jones delivered an invited seminar today at the National Institutes of Health (NIH) / National Eye Institute (NEI) today on Retinas, How Are They Wired? How Does Wiring Change In Disease?
Large-Scale Transmission Electron Microscopy and Connectomics
Synapses, Gap Junctions, Adherens, and Tight Junctions, Oh My!
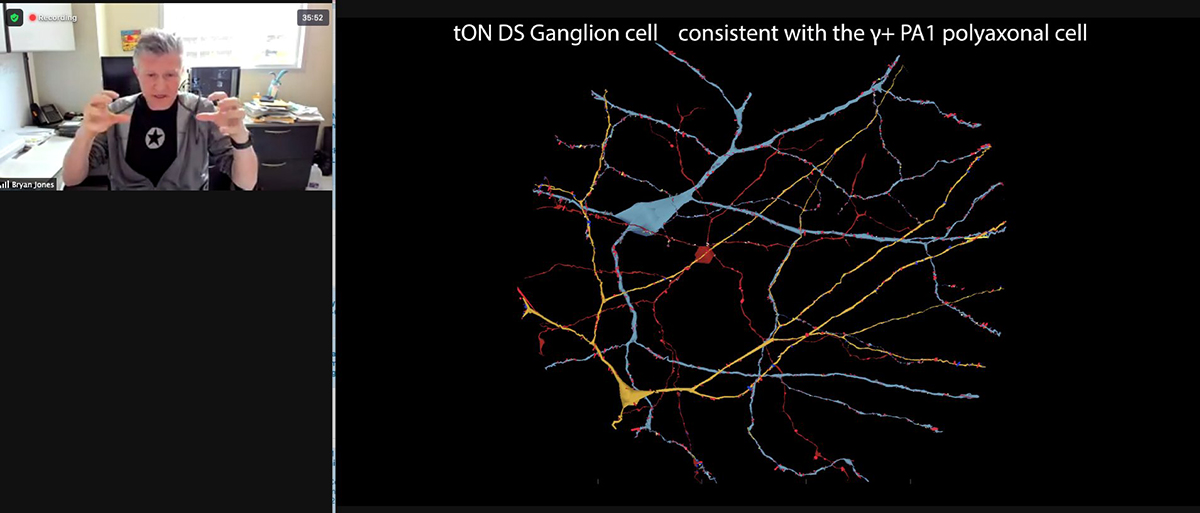
PI, Bryan William Jones gave a talk yesterday to the NSF NeuroNex working group on Synapses, Gap junctions, Adherens, and Tight Junctions and their role in connectomics.
Will post the video of that talk here, if and when it becomes available.
Thanks to friend and colleague Uri @manorlaboratory for screenshotting in the middle of the talk.
Selena Sullivan, Outstanding Undergraduate Researcher Award
Congratulations to Selena Sullivan, an undergraduate in the laboratory for earning the Outstanding Undergraduate Researcher Award at the University of Utah, School of Medicine for 2021.
ARVO 2021
PI, Bryan William Jones gave a talk at ARVO yesterday, in a special session on retinal degeneration and plasticity, unfortunately virtually, but it is the right thing to do in a pandemic. That makes effectively two years in a row where we have not traveled to see colleagues at our vision meetings. That said, the meetings are still productive.
Laboratory In A Pandemic
This past year has been remarkable in terms of the impact that COVID-19 has wrought, as well as its impact upon everyone in the team. I am proud of the resiliency and huge efforts that people have gone through to keep science in the lab going, and cannot possibly relate how grateful I am to everyone. They have struggled through working remotely, human resources problems, child-care issues, logistical issues related to access of research tools and data, equipment downtime due to maintenance issues, personal COVID infections, and outbreaks in their families, not being able to attend meetings, duties to the department and university to assist with the COVID-19 response, volunteering of time, money and resources to assist the COVI-19 response, having to be responsive to new IT demands, and so much more.
I had been tracking COVID-19 since January, but by March, I became particularly alarmed and shut the lab down, sending everyone home with computers in short order. Having no idea how this would turn out, I did have visions of being able to get all caught up and do a ton of reading that I’ve not been able to do, and more. 2020 being 2020, that was just not possible, and the bureaucratic overhead of reporting and constant emails and videoconferencing has eaten up massive amounts of time. This year, despite the global pandemic, we’ve still managed to publish 6 manuscripts, 3 pre-prints, 2 abstracts, and 1 chapter, and secured a new NSF grant. We’ve continued to mentor graduate students, undergraduate students, and post-docs. Again, I am incredibly proud of this team.
The danger with all this productivity in the face of a global pandemic is that we are burning our team out. So, the plan was to take a couple weeks off, spend time with the closest people in our lives, recharge our batteries, and hit the ground running again in the New Year. Reality intruded for me at least however, and I discovered a water leak from one of the electron microscope chillers that leaked into the walls. I am investigating this now, but we are clearly down with half of our ultrastructural infrastructure unavailable until we can either get the chiller/microscope repaired or equipment replaced. 2020 just will not let go… The lab is still working from home, but I’ve had to go into work and get some things done related to the water leak.
Outside of equipment failures and water leaks, working in the lab during 2020 has been a challenge. After getting approval to have some people return to campus, we decided to bring technicians back for part time work in the lab and part time work at home, so long as they could keep distances while wearing masks while in the lab. Not being able to be in the lab full time, and having to maintain physical distance from all others and wear masks and personal protective gear constantly has absolutely been an added burden. However, we are fortunate in that we have space to spread out, but it has still been a massive challenge, getting work done.
Our lab has helped with the COVID-19 response by taking temperatures and performing health screenings of visitors and patients/visitors to the Moran Eye Center, a task all of our labs in the Moran Eye Center have been participating in. It takes us away from our research duties, but also helps in some small way with the COVID-19 response.
Staying up on the work from students and post-docs has, like everything else, gone virtual. This slows everything down of course and contributes to the sense of isolation. Both Crystal @CSigulinsky and Becca @BeccaPfeiffer19 have been critical in this mentoring effort. I can’t wait until we get this thing back under control and can meet in person again…
I’m also grateful for Mark Kirkpatrick, and Kevin Mcilwrath from JEOL @JEOLUSA who helped us maintain the microscopes and keep them up and running. And when they went down, I am so grateful that Kevin could come in and help us get them back up and running. We definitely had downtime as a result of the pandemic, but we would have had more, if it were not for Kevin Mcilwrath. We will need him again, to get back up and running after the Christmas Eve chiller failure, and I am grateful for him and JEOL for making the efforts to keep us running.
We’ll see what the New Year brings. The list of things to do starting in a few days is formidable. Even though the vaccines are just now starting to roll out, we are still in the middle of a pandemic and will not be returning to normal functioning in the lab for months yet. We have to perform repairs to our infrastructure, finish a manuscript I am working on, edits to a colleagues manuscript, getting a couple of manuscripts from post-docs going, a couple of new genetic models to create, a grant renewal to write, data to process for collaborations, getting our light microscopes back up and running, a grad student starting a rotation, undergrads presenting their research, and then completing work on all the other stuff that we normally have to do.
I am so grateful for Jia-Hui, Jamie, Hope, Nat, Crystal, Becca, Jeebika, Jessica, Selena and Olivia. You all made getting through this year possible. Thank you for your teamwork, and your *hard* work. Also, my undying gratitude to my chairman, and all the colleagues in my department. What a brutal year, and my hopes are for more normalization, better leadership at the federal level, and mitigation of the COVID-19 pandemic as vaccines start to roll out.
Happy Weird Holidays, 2020 From The Marclab For Connectomics
Happy Weird Holidays, 2020 to you all from us here at the Marclab for Connectomics.
What a strange, and fundamentally challenging year this has been for so many. It has been lonely, frustrating, painful, and discouraging for many of us, and I encourage all to be a bit more patient, kind, and compassionate to those around you, as you don’t know what they are dealing with.
This pandemic has exacerbated the challenges that many are dealing with financially, socially, and emotionally. It has led to incredible bureaucracy for many of us trying to run labs. Personnel issues as folks in labs struggle with the issues in their lives not only working from home, but working from home during a pandemic, including child care, or care for other family members. Struggles with personal illness (COVID and otherwise). Financial issues related to employment. Struggles with depression and isolation. Inability to collaborate, or attend meetings that are critical to ongoing projects. Loss of research resources. Inability to get equipment or service on equipment… For some, this pandemic has resulting in a complete cessation of research activities.
Lab productivity has certainly been hugely impacted for many labs and the implications of that for their NIH or NSF funding is still unclear. Our productivity has certainly suffered this year, and we are absolutely behind on promised data generation. I don’t know how we are going to deal with that yet. The goal is to try and catch up, and while our lab is nominally working right now, all undergraduate, and graduate students are working remotely. Postdocs are partially working in the lab, and technicians are splitting their time. Until all the vaccines roll out, it is unclear how much longer this will have to continue. Fortunately for us, we are heavily computational and we’ve been able to still be productive on existing data. We also have generous lab space that we can spread out in for those who are working on campus. So, for now, we are forging ahead, managing as best as we can and attempting to be helpful to others where we can by processing tissues for other labs where we can to help them with their productivity problems for instance, and as always, reviewing manuscripts and grants in-between trying to get our manuscripts and grants in.
This pandemic has exacerbated the challenges that many are dealing with financially, socially, emotionally, and scientifically. And while this life gives and it takes, it also gives us opportunities to make decisions that allow us to help improve the lives of others, and be a force for good.
Happy Holidays from the Marclab for Connectomics.