This abstract was presented today, April 24th at the 2023 Association for Research in Vision and Opthalmology (ARVO) meetings in New Orleans, Louisiana by Taylor Otterness, Crystal Sigulinsky, James Anderson, and Bryan William Jones.
Full resolution version here.
Purpose
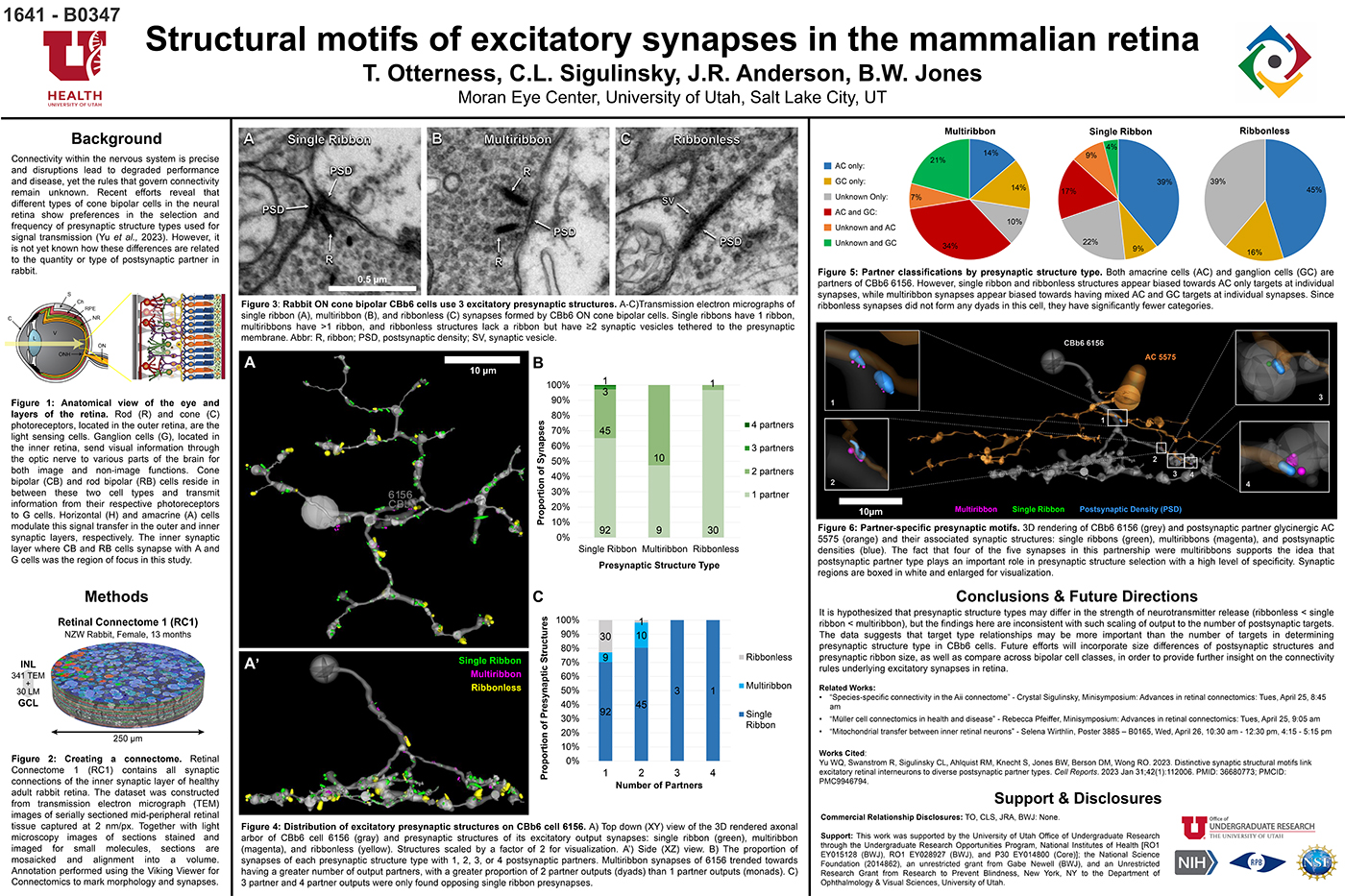
Connectivity within the nervous system is precise and disruptions lead to degraded performance and disease, yet the rules that govern connectivity remain unknown. Recent efforts reveal that different types of cone bipolar cells in the neural retina show preferences in the selection and frequency of presynaptic structure types used for signal transmission. However, it is not yet known how these differences are related to the quantity or type of postsynaptic partner. We used Retinal Connectome 1 (RC1) to analyze the synaptic output of rabbit CBb6 cells, a type of ON cone bipolar cell that forms excitatory synapses via diverse presynaptic structure types, to identify patterns in how these cells interact with their postsynaptic partners.
Methods
RC1 is a 0.25 mm diameter volume sampled from mid-peripheral retina of a 13 month old female Dutch-Belted rabbit, serially sectioned at 70 nm, and imaged at ultrastructural resolution (2nm/px) using transmission electron microscopy. Postsynaptic partners of CBb6 cell 6156’s presynaptic structures were annotated using the Viking Viewer for Connectomics. Statistical analyses were conducted in Microsoft Excel and investigated further with 3D rendering and graph visualization of connectivity.
Results
The factors tracked for comparison included presynaptic structure type, target number, and postsynaptic partner type. Multiribbon synapses of CBb6 cell 6156 trended towards having a greater number of output partners, with a greater proportion of dyads than monads. Despite this, triads and quadrads were only found opposing single ribbon synapses. As the different presynaptic structure types may differ in the strength of neurotransmitter release (ribbonless < single ribbon < multiribbon), these findings are inconsistent with scaling of output to the number of postsynaptic targets. Both amacrine cells (AC) and ganglion cells (GC) are postsynaptic partners of 6156. However, single ribbon and ribbonless structures appear biased towards AC only targets, while multiribbon synapses appear biased toward mixed AC and GC targets.
Conclusions
Target type relationships appear more important than the number of targets in determining presynaptic structure type in CBb6. Future efforts will examine size differences of postsynaptic structures and presynaptic ribbon size, and even compare across bipolar cell classes, in order to provide further insight on the connectivity rules underlying excitatory synapses.