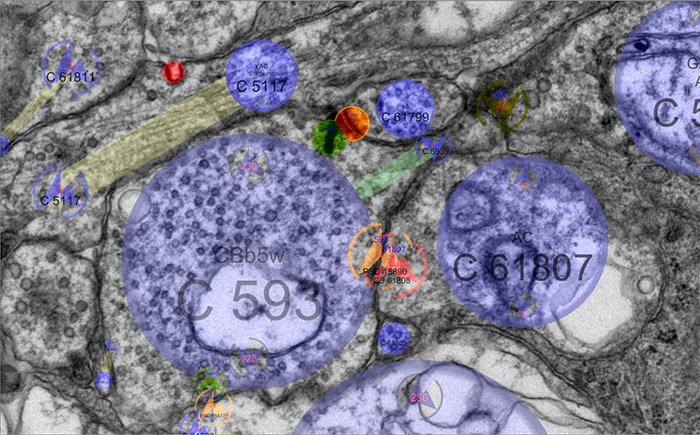
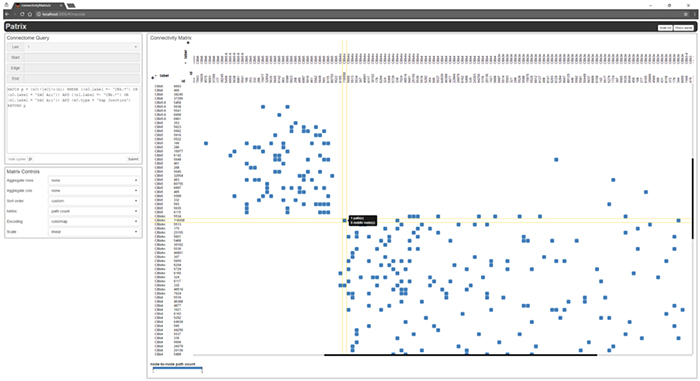
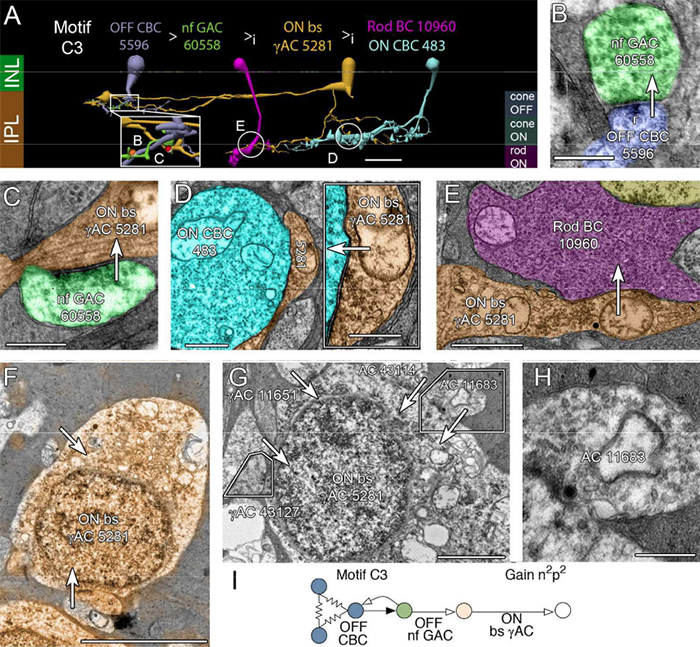
We presented a poster on Mapping the network architecture of gap junctional coupling among parallel processing channels in the mammalian retina at the 2019 HHMI Connectomics meeting in Berlintoday. Downsampled PDF of poster here.
Authors: Crystal L. Sigulinsky, James R. Anderson, Ethan Kerzner, Christopher N. Rapp, Rebecca L. Pfeiffer, Daniel P. Emrich, Kevin D. Rapp, Noah T. Nelson, J. Scott Lauritzen, Miriah Meyer, Robert E. Marc, and Bryan W. Jones.
Abstract: Electrical synapses are fundamental components of neural networks. Gap junctions provide the anatomical basis for electrical synapses and are prevalent throughout the neural retina with essential roles in signal transmission. Gap junctions within and between the parallel processing channels afforded by retinal bipolar cells have been reported or predicted, but their roles, partners, and patterns remain largely unknown. Here, we took advantage of the high resolution of Retinal Connectome 1 (RC1) to reconstruct ON cone bipolar cells (CBCs) and map their coupling topologies.