We have a new manuscript out in Clinical Neurophysiology, An Update on Retinal Prostheses. PubMedDirect Link PDF here.
Authors: Lauren N. Ayton, Nick Barnes, Gislin Dagnelie, Takashi Fujikado, Georges Goetz, Ralf Hornig, Bryan W. Jones, Mahiul M.K. Muqit, Daniel L. Rathbun, Katarina Stingl, James D. Weiland, Matthew A. Petoe.
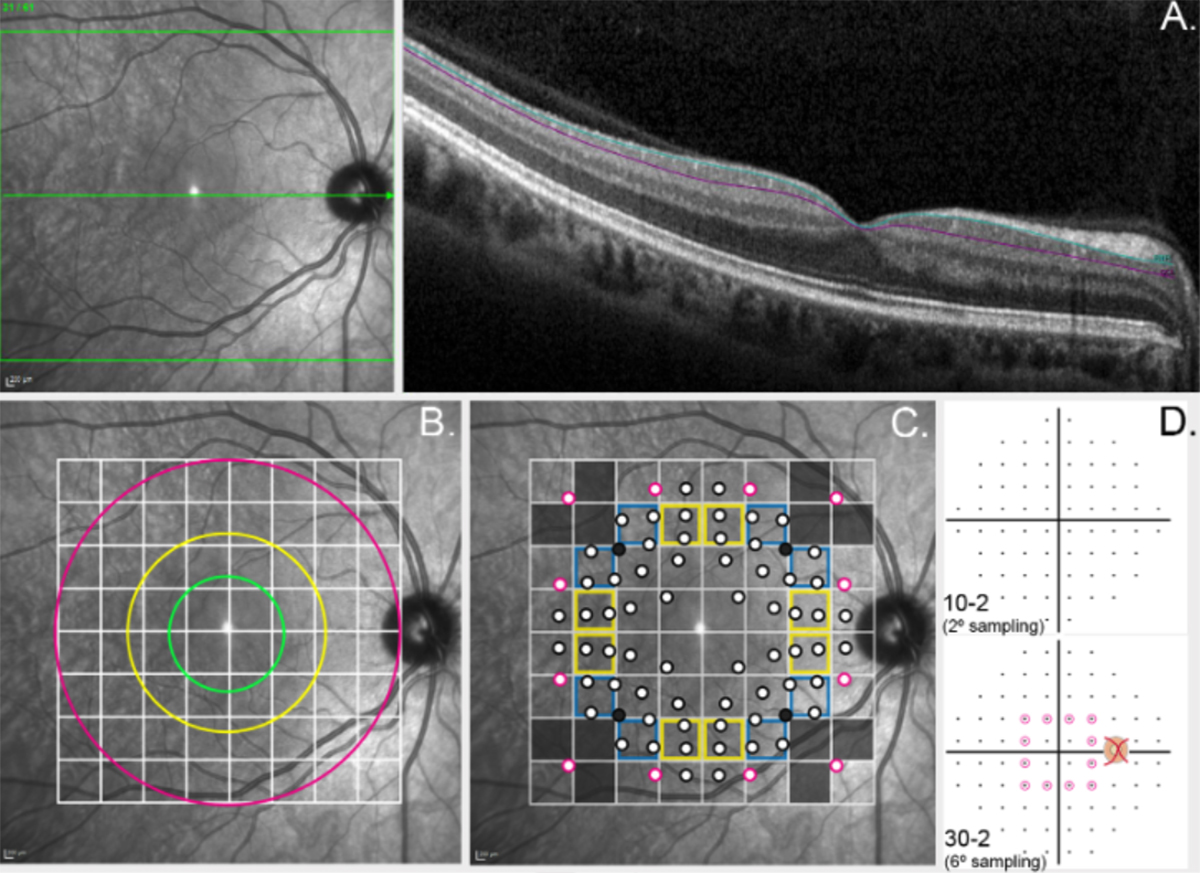
Abstract: Retinal prostheses are designed to restore a basic sense of sight to people with profound vision loss. They require a relatively intact posterior visual pathway (optic nerve, lateral geniculate nucleus and visual cor- tex). Retinal implants are options for people with severe stages of retinal degenerative disease such as retinitis pigmentosa and age-related macular degeneration.
There have now been three regulatory-approved retinal prostheses. Over five hundred patients have been implanted globally over the past 15 years. Devices generally provide an improved ability to localize high-contrast objects, navigate, and perform basic orientation tasks. Adverse events have included con- junctival erosion, retinal detachment, loss of light perception, and the need for revision surgery, but are rare. There are also specific device risks, including overstimulation (which could cause damage to the retina) or delamination of implanted components, but these are very unlikely.
Current challenges include how to improve visual acuity, enlarge the field-of-view, and reduce a com- plex visual scene to its most salient components through image processing. This review encompasses the work of over 40 individual research groups who have built devices, developed stimulation strategies, or investigated the basic physiology underpinning retinal prostheses. Current technologies are summarized, along with future challenges that face the field.